|
2/19/2024 0 Comments Metal reactivity definition
After corrosion had occurred clean the corroded surface and again measure the mass of the metal block.Metals are substances that have high electrical conductivity, high thermal conductivity, and high density, to name a few characteristics. To measure the mass lost, weigh a clean pure piece of metal and then expose it to the environment for a definite period. Hence, to measure how much corrosion occurred one should measure the mass lost from the metal. The answer is the mass of the metal is lost. Since corrosion lead to the loss of metal, one can ask what is lost. With the help of this model, we can predict and control the formation of Oxide layers even in adverse situations. The formation of an Oxide layer on metal is explained by the Deal Grove model. Temperature: The rate of Corrosion increases with temperature. Humidity in Weather: When the weather is humid, the presence of moisture with a high temperature accelerates corrosion because ions gain energy and begin moving quicker in a higher temperature, causing them to collide more frequently. An anode is a location with less oxygen concentration, whereas a cathode is a region with a higher oxygen concentration. Corrosion is accelerated when electrolytes are present in water.Ĭoncentration of Oxygen: Corrosion is accelerated by an increase in oxygen content. Presence of Electrolytes: Because they carry ions, dissolved salt in water acts as an electrolyte. Impurities in Metals: In general, the presence of contaminants in metals accelerates corrosion because these impurities operate as the microscopic electrochemical cell that causes corrosion. Iron, for example, is quickly oxidised, but gold is not because gold is less reactive than iron. Reactive metals lose electrons more quickly and corrode more quickly. Position of Metals in Electrochemical Series: In corrosion, the electrochemical series is extremely important. Various factors influencing the corrosion of any material are, The most common exception to this is the metal Aluminium as even though it is placed up in the periodic table then also it does not corrode easily. metal like iron and copper corrode very easily, whereas metal like gold and platinum does not corrode at all or corrode very little. So all metal can be corroded but their rate of corrosion varies a lot. In the reactivity series, the metal that is placed up corrodes very easily than the metal placed below in the reactivity series. The elements placed higher up in the reactivity series is more reactive as compared to the element placed below the element in the reactivity series. Do All Metals Corrode?Ī reactivity series is a series of elements based on their reactivity. Corrosion is a redox reaction that causes the loss of pure metal by converting them into oxide and thus decreasing its strength and other physical properties. The degradation of pure material when they react with moisture, air or acid is called Corrosion. Software Engineering Interview Questions.Top 10 System Design Interview Questions and Answers.Top 20 Puzzles Commonly Asked During SDE Interviews.Commonly Asked Data Structure Interview Questions.Top 10 algorithms in Interview Questions.Top 20 Dynamic Programming Interview Questions. 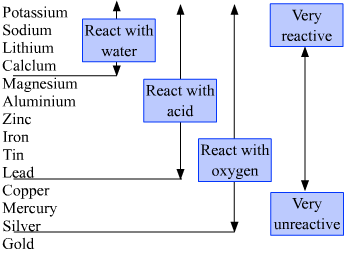
Top 20 Hashing Technique based Interview Questions.Top 50 Dynamic Programming (DP) Problems.Top 20 Greedy Algorithms Interview Questions.Top 100 DSA Interview Questions Topic-wise.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
